Description
Intact Genomics T4 DNA Ligase catalyzes the formation of a phosphodiester bond between juxtaposed 5’-phosphate and 3’-hydroxyl termini in duplex DNA or RNA. This enzyme joins DNA fragments with either cohesive or blunt termini and repairs single stranded nicks in duplex DNA, RNA or DNA/RNA hybrids (1).
Applications
• Cloning of restriction enzyme generated DNA fragments
• Cloning of PCR products
• Next-gen library preparation
• Joining linkers and adapters to cohesive or blunt-ended DNA
• Nick repair in duplex DNA, RNA or DNA/RNA hybrids
• Self-circularization of linear DNA
Purity
The physical purity of Intact Genomics T4 DNA Ligase is ≥99% as assessed by SDS-PAGE with Coomassie® blue staining (see figure below).

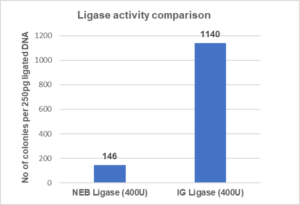
Comparison Testing
Intact Genomics T4 DNA displays significantly higher ligation efficiency than the nearest competitor.

Product Source
E. coli strain expressing a recombinant clone
Product Includes
- T4 DNA Ligase
- 10x T4 DNA Ligase Reaction Buffer
Storage Buffer
50 mM Tris-HCl, 50 mM KCl, 1 mM DTT, 0.1 mM EDTA, 50% Glycerol, pH 7.5 @ 25 ºC
1x T4 DNA Reaction Buffer
50 mM Tris-HCl, 10 mM MgCl2, 10 mM DTT, 1 mM ATP, pH 7.5 @ 25 ºC
Unit Definition
One unit is defined as the amount of enzyme required to give 50% ligation of HindIII fragments of λ DNA (250 ng/µl) in a total reaction volume of 20 μl in 30 minutes at 16°C in 1X T4 DNA ligase reaction buffer.
Inhibition and Inactivation
- Inhibitors: metal chelators, phosphate and ammonium ions, KCl and NaCl at a concentration higher than 50 mM.
- Inactivated by heating at 70 °C for 15 min or by addition of EDTA.
Quality Control Assays
- Endonuclease Activity (Nicking)
1 µg of supercoiled plasmid DNA is incubated with 20 units of T4 DNA Ligase in 1x Ligase buffer for 2 hours at 37 ºC. Following incubation, the supercoiled DNA is visualized on an ethidium bromide-stained agarose gel. No visible nicking or cutting of DNA was found.
- Functional Assay
DNA Ligase functional efficiency is tested in cloning assays.3212 3216 3217